The most important quality parameter for gasoline is the octane quality. Octane number is a measure of the antiknock properties of the fuel. Knocking in a gasoline engine is a metallic clattering noise (pinging), which indicates excessive intensity in preflame reactions. Severe knocking can damage the engine.
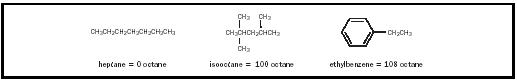
Preflame reactions occur in the engine cylinders when portions of the fuel self-initiate combustion prior to the advancing flame from the spark plug. This additional combustion causes an excessive rate of energy release, which is knock. The tendency of a fuel to engage in preflame reactions is dependent upon the structure of its component molecules (see Figure 1);
the tendency for preflame reactions is high for straight chain hydrocarbons, medium for branched hydrocarbons, and low for aromatics.
The octane number for a test gasoline represents the percentage by volume of isooctane (2,2,4-trimethylpentane) in a reference fuel consisting of the mixture of isooctane and heptane that would be necessary to match the test fuel’s knocking tendency. Isooctane burns with a minimal knocking and is given an octane rating of 100. This is in contrast to heptane, which burns with much knocking and is given an octane rating of 0. Thus, a gasoline that burns with the same amount of knocking as a mixture of 92 percent isooctane and 8 percent heptane is classified as a 92 octane gasoline.
The octane ratings of gasoline can be increased by the addition of small amounts of antiknock agents. The first commercially successful antiknock agent, tetraethyllead (TEL), was developed in the 1920s. TEL was used to promote the development of higher efficiency, higher compression engines. However, TEL is highly toxic and poisons catalytic converters. Since 1974 all new U.S. automobile engines have used catalytic converters in order to reduce exhaust emissions.
Methyl t -butyl ether (MTBE) has been the antiknock agent of choice for unleaded gasoline. MTBE provides high-octane quality along with low volatility and is readily soluble in gasoline. However, leakage of gasoline from underground storage tanks has resulted in the detection of MTBE in the drinking water of several urban areas. This prompted the state of California to order the removal of MTBE from California gasoline by 2003.
Alcohols also have found use as octane enhancers. At higher concentration alcohols can be used as gasoline extenders, thus decreasing our dependency upon imported crude oil. A significant portion of all U.S. marketed gasoline is believed to contain ethanol.